
T4. ‼️ FDA-approved ≠ FDA-cleared
This is a common error I see in papers, conferences, and webinars. Most radiology AI applications are FDA-cleared, not FDA-approved.
Learn more 👉 buff.ly/43UDVHR
#RadAIchat


T2: I personally love this explorer: artificialintelligenceact.eu/ai-act-explore…
It allows you to search anything specific within this heavy document.
Save for later!
#radAIchat


Radiology: Artificial Intelligence T4. This paper reviews process and challenges for differenft FDA algorithms pathways.pubs.rsna.org/doi/abs/10.114…
#RadAIChat

This work released in oct 2023 World Health Organization (WHO) serves as a global inspiration and basis for all regulations.
key topics:
1. Documentation and transparency
2. Risk management and AI systems development lifecycle approaches
3. Intended use, and analytical and clinical validation
#RadAIchat





T4: again, risk based approach is key.
One of the things different from the Medical Device Regulation is more stringent monitoring.
Can you name any more differences? (there are many)
#RadAIChat

2/ T1.
2️⃣ Addressing bias and fairness
3️⃣ Protecting patient privacy and ensuring data governance and security
4️⃣ Promoting transparency
5️⃣ Establishing accountability
6️⃣ Building public trust
7️⃣ Balancing innovation and risk
#RadAIchat


This concludes today's #RadAIchat . Many thanks to our experts and participants for the thought-provoking discussion. Our panelists will continue to engage with participants 🌍 for the next 24 hours. Join us for the next #RadAIchat on Jun. 5, 2024 at 8 PM ET.


1/ T1. Medical AI should be regulated for several reasons:
1️⃣ Ensuring safety and efficacy: #MedicalAI can have significant impacts on patient outcomes, so they must be thoroughly tested and proven to be safe and effective before being deployed. 🧵
#RadAIchat


Here is a sneak peek at tonight's #RadAIchat at 8 PM ET on 'Medical #AI Regulation: A Primer & Future Directions' moderated by Merel Huisman MD PhD Tugba Akinci D'Antonoli and panelists Dania Daye, MD PhD James Hillis Woojin Kim #radiomics #AI #DL #radres RSNA Charles Kahn, MD Hesham Elhalawani


Welcome to Radiology: Artificial Intelligence chat on 'Medical #AI Regulation: A Primer & Future Directions' moderated by Merel Huisman MD PhD Tugba Akinci D'Antonoli and panelists Dania Daye, MD PhD James Hillis Woojin Kim RSNA Charles Kahn, MD Hesham Elhalawani
Please introduce yourself and always remember to add #RadAIchat



T4. The #FDA adopts a risk-based approach, and different pathways are available depending on risk, such as premarket approval, de novo or 510(k) #RadAIchat Radiology: Artificial Intelligence

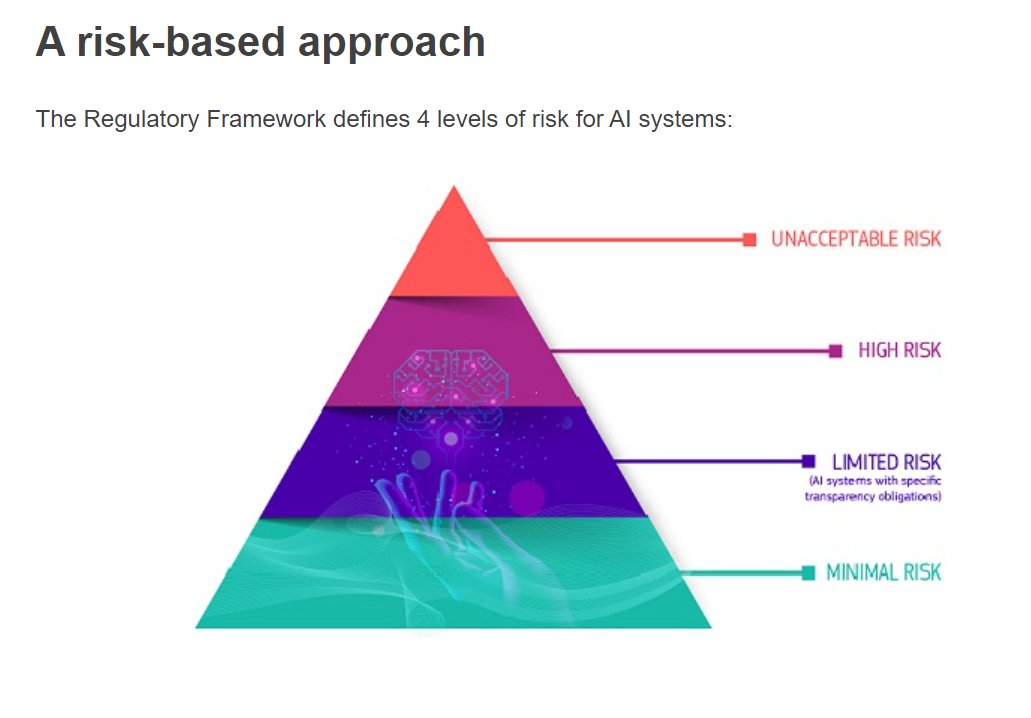
T2: The EU AI Act will come into force soon.
A key concept is the risk-based approach.
All medical AI = high risk
#radAIchat
follow Hugh Harvey for updates



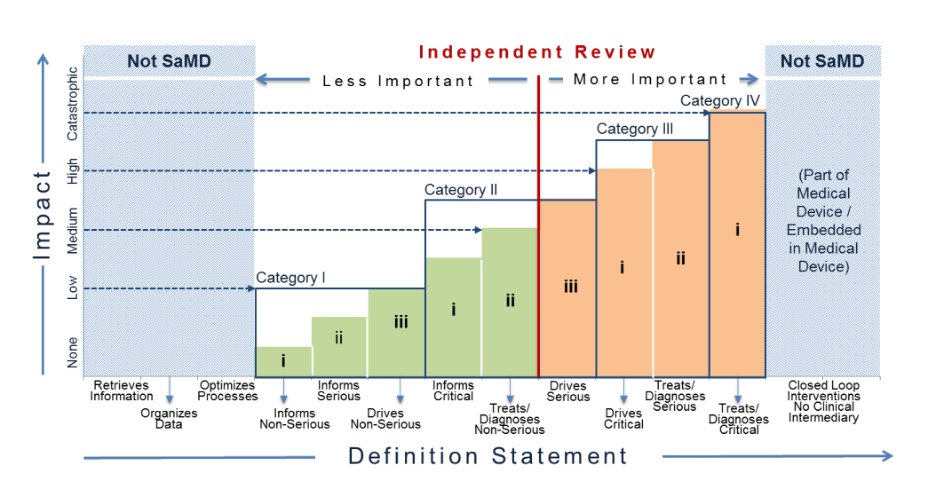
T3. Published in 2021, this article highlights some of the differences as well.
#RadAIchat #AItransparency
buff.ly/3zcvM1y






