
Dr Anup Amatya FDA Oncology gives a masterclass on subgroup analysis as introduction to panel 4
Must consider
- sample size / power
- multiplicity
- biological rationale
Should be done when there is persuasive evidence of benefit in overall population
#AACRSciencePolicy


Did you miss the 7/18/2023 FDA-AACR-ASA Workshop on Overall Survival in #Oncology #ClinicalTrials ? Slides & recording are now available:
aacr.org/professionals/…
#AACRSciencePolicy #regulatory #biostats #drugdevelopment #OncTwitter


Love catching Yale Cancer Center being repped, often by Roy Herbst like at today's U.S. FDA -AACR Workshop on How Much Is Enough? Trial Designs for Treatment Regimens with Multiple Phases. I appreciate the focus on the benefit possible through improved design #AACRSciencePolicy


Great to meet Dr Kamal Menghrajani, MD, MS, haematologist and leukaemiologist currently working at the The White House and chat about the intersection of haematology & public health (& the MORPHO trial results) #AACR #AACR 24 #aacrsciencepolicy


.U.S. FDA leaders participate in a Fireside Chat at the FDA-AACR Workshop on Trial Designs for Treatment Regimens with Multiple Phases. FDA Oncology #AACRSciencePolicy


AACR Past President Elizabeth Jaffee moderates this session on Optimizing Perioperative Treatment Regimens at the FDA-AACR workshop on trial design. Jhanelle Gray MD of Moffitt Cancer Center introduces the session. #AACRSciencePolicy Dr. Elizabeth Jaffee @HopkinsKimmel


AACR Past President Elizabeth M. Jaffee and @FDAoncology's Harpreet Singh welcome participants to this joint workshop on Trial Designs for Treatment Regimens with Multiple Phases:
bit.ly/3QDI4eL #AACRSciencePolicy Dr. Elizabeth Jaffee



Our panel of experts discusses the landscape for perioperative trial designs: Anup Amatya, Valsamo Anagnostou, Paz Vellanki, Thelma Brown, Roy Herbst, Mark Kris, and Craig Tendler. #AACRSciencePolicy U.S. FDA @HopkinsKimmel Translational Breast Cancer Research Consortium Roy Herbst Yale Cancer Center Memorial Sloan Kettering Cancer Center


How Much is Enough? Trial Designs for Treatment Regimens with Multiple Phases:
AACR Past President Dr. Elizabeth Jaffee and Harpreet Singh will chair this AACR-U.S. FDA workshop. Join us online May 9. Learn more:
bit.ly/3JULR3I
#AACRSciencePolicy Harpreet Singh, MD



The Cancer Moonshot: Opportunities to Fulfill the Vision of the National Cancer Plan—Roy Herbst moderated an #AACRSciencePolicy session today at #AACR24 , featuring Dr. Kimryn Rathmell Kimryn Rathmell, AACR President Patricia M. LoRusso Patricia M. LoRusso, Dr. Elizabeth Jaffee, and Bianca Islam, MD, PhD, MSc (she/her).



- flags importance of considering harm signal if seen across multiple studies (often FDA Oncology uniquely positioned to see this as it has all the data)
- if you have a biomarker-defined population where expect differential effect, should prespecify analysis
#AACRSciencePolicy


Keith Flaherty Mass General Cancer Center summarizes the panel's discussions
- even nonsignificant trends in OS harm warrant consideration
- look at evidence of toxicity / AEs as evidence of potential harm, especially if not anticipated
#AACRSciencePolicy


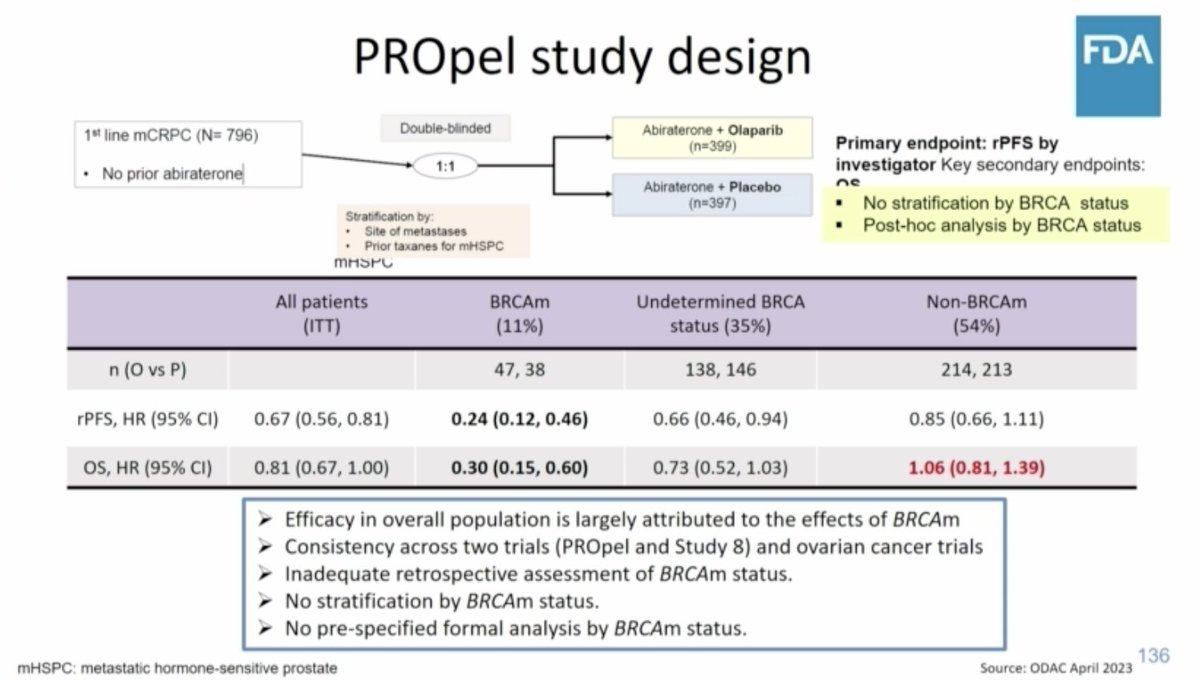
Uses the example of PROpel & olaparib in metastatic castrate-resistant prostate cancer (mCRPC) & recent ODAC meeting
- efficacy in overall pop largely due to effects in BRCAmut pts
- indication narrowed to only be approved for BRCAmut patients
#AACRSciencePolicy


How Much is Enough? Trial Designs for Treatment Regimens with Multiple Phases:
AACR Past President Dr. Elizabeth Jaffee and Harpreet Singh will chair this AACR-U.S. FDA workshop. Join us online May 9. Learn more:
bit.ly/3Us9fu3
#AACRSciencePolicy Harpreet Singh, MD


Harpreet Singh of FDA Oncology provided closing remarks for the U.S. FDA-AACR Workshop on Trial Designs for Treatment Regimens with Multiple Phases. Recordings and slides will be posted to the AACR website by May 23. bit.ly/44yTJB8 #AACRSciencePolicy


“..it’s unacceptable for patients to experience chronic low grade nausea or diarrhea..” Dr. LoRusso Yale Cancer Center. Excellent presentations & possible ways to improve #doseoptimization by #AACRSciencePolicy FDA Oncology
#bcsm
the.right.dose




